Indian Pharmaceutical Industry: From Pre-2000 Foundations to the Post-COVID Era
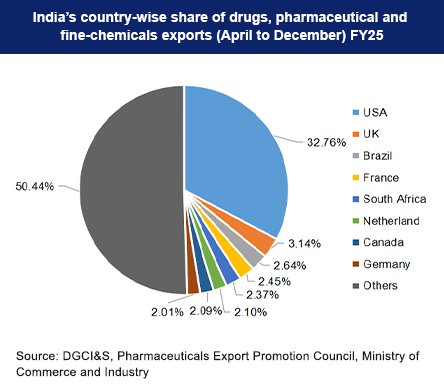
The Indian pharmaceutical industry is one of the most transformative success stories in global healthcare. From being a largely import-dependent sector before 2000 to becoming the “Pharmacy of the World” after COVID-19, India’s pharma journey reflects resilience, innovation, and scale.
At Pharmakens Healthcare, we believe understanding this evolution is crucial—not just for industry professionals, but for anyone invested in the future of healthcare.
This blog explores the Indian Pharma Industry across three major phases:
Pre-2000 Era
2000 to Pre-COVID Period
Post-COVID Transformation
1. Indian Pharmaceutical Industry: Pre-2000 Era (Before Globalization)
Limited Beginnings, Strong Intent
Before 2000, India’s pharmaceutical industry was primarily domestic-focused and heavily influenced by government regulation.
Key Characteristics
Process Patent Regime (1970 Patent Act)
India allowed only process patents, not product patents. This enabled companies to reverse-engineer medicines and produce affordable generics.Import Dependency
APIs (Active Pharmaceutical Ingredients) and advanced formulations were largely imported, making India dependent on global suppliers.Focus on Essential Medicines
Production centered on antibiotics, painkillers, vitamins, and basic formulations.Low R&D Investment
Innovation was limited. Most companies focused on cost-effective manufacturing rather than drug discovery.
Early Industry Contributors
Indian companies began laying strong foundations during this period, including Sun Pharmaceutical Industries, Cipla, and Dr. Reddy’s Laboratories, which later became global leaders.
👉 Strength of this era: Affordable medicines for India
👉 Limitation: Limited global presence and innovation
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
2. Indian Pharma Industry: 2000–2019 (Global Expansion Phase)

Liberalization & Global Recognition
The post-2000 era marked India’s entry into the global pharmaceutical ecosystem.
Major Turning Points
TRIPS Agreement (2005)
India aligned with WTO’s TRIPS agreement, reintroducing product patents. This forced companies to:
Increase R&D
Improve compliance
Focus on quality and innovation
Rise of Generics Exports
Indian companies became leaders in generic medicines, especially in:
USA
Europe
Africa
Latin America
India emerged as:
Largest supplier of generic drugs globally
Supplier of 40% of US generics
Regulatory Excellence
Indian manufacturing plants received thousands of approvals from:
US FDA
UK MHRA
WHO-GMP
Companies like Lupin and Aurobindo Pharma expanded aggressively into regulated markets.
Growth of Contract Manufacturing
India became a hub for:
Contract Research (CRO)
Contract Manufacturing (CMO)
Clinical trials
👉 Strength of this era: Global trust & export leadership
👉 Challenge: API dependency on China
3. Indian Pharma Industry During COVID-19 (2020–2021)

India as the Global Healthcare Lifeline
COVID-19 was the most defining moment for Indian pharma.
Key Contributions
Vaccine Leadership
India became the largest vaccine manufacturer during the pandemic.
The world relied on Indian vaccines and medicines for survival.
Uninterrupted Medicine Supply
Despite lockdowns:
Indian pharma ensured continuous supply of essential medicines
Exported life-saving drugs to over 200 countries
Rapid Innovation
Indian companies scaled production of:
Antivirals
Antibiotics
Oxygen-related medicines
Global Recognition
India earned the title “Pharmacy of the World” due to:
Speed
Scale
Affordability
👉 Strength of this era: Trust, responsibility, and resilience
👉 Lesson learned: Supply chain self-reliance is critical
4. Indian Pharma Industry: Post-COVID Era (2022 Onwards)
A Shift Towards Innovation & Self-Reliance
Post-COVID, Indian pharma entered a new strategic phase.
Major Trends
API & Bulk Drug Self-Reliance
Government initiatives like PLI schemes encouraged:
Domestic API manufacturing
Reduced dependence on imports
Focus on Innovation
Companies are now investing in:
Biosimilars
Specialty medicines
Oncology and chronic therapies
Digital Transformation
Pharma operations adopted:
AI in drug discovery
Digital quality management
Supply chain automation
Sustainability & Compliance
Environmental responsibility, ESG norms, and ethical manufacturing are now core priorities.
👉 Strength of this era: Innovation + sustainability
👉 Future focus: Quality, research, and global leadership
5. The Road Ahead: Future of Indian Pharma

The Indian pharmaceutical industry is poised to become:
A global innovation hub
A leader in complex generics and biologics
A benchmark for affordable and ethical healthcare
With strong government support, skilled talent, and manufacturing excellence, India’s pharma sector is entering its most powerful phase yet.
Conclusion: Pharmakens Healthcare Perspective
At Pharmakens Healthcare, we see the Indian pharmaceutical industry not just as a business ecosystem, but as a lifeline for global health.
From humble beginnings before 2000 to becoming a post-COVID global leader, Indian pharma’s journey is a story of vision, adaptability, and responsibility.
As the industry evolves, Pharmakens Healthcare remains committed to:
Quality excellence
Ethical practices
Sustainable healthcare solutions
Together, we move forward—stronger, smarter, and healthier.
